
Biosafety
Institutional Biosafety Committee (IBC)
The Institutional Biosafety Committee provides oversight to ensure that all research involving recombinant or synthetic nucleic acids, infectious agents, and other biohazardous materials is conducted safely and in compliance with federal, state, and institutional requirements. As mandated by the NIH Guidelines, the IBC reviews and approves applicable research protocols, assesses potential risks, and ensures that appropriate containment practices and facilities are in place.
Through its review, guidance, and ongoing monitoring, the IBC promotes a safe research environment, supports responsible scientific innovation, and helps safeguard the health of researchers, the campus community, and the public.
General Safety Information
 General Laboratory Safety Manual
General Laboratory Safety Manual
 SciSchield
SciSchield
 BMBL 6th Edition
BMBL 6th Edition
 Dual Use Research of Concern
Dual Use Research of Concern
IBC Resources
Biosafety and Compliance Issues in Research Laboratories
Use of Biohazardous Materials and Recombinant or Synthetic Nucleic Acid
Molecules in Research
Principal Investigators are responsible for ensuring IBC review of the following:
- Animal experiments requiring IBC approval with any of the above (prior to IACUC approval)
- Biological Toxins (Select Agent toxins below the permissible toxin amount)
- Human and Non-human Primate (NHP): blood, body fluids, tissues and primary or established cell lines
- Pathogens/Microorganisms: Risk Group 1 or higher
- Recombinant or Synthetic DNA work as defined by the NIH Guidelines
Most importantly, the IBC’s objective is to ensure that such activities meet standards of good biological safety practices, emphasizing protection of personnel, the public, and the environment.
All communication should be directed to the IBC office at: ibc@westernu.edu
WesternU IBC utilizes CITI for all compliance related training. Please follow the instructions for registration and completion of IBC required courses though CITI.
IBC related forms can be found in the IRBnet Forms/templates library www.irbnet.org under Western University of Health Sciences IBC – Documents for Researchers. For questions about IBC forms or IRBNet please contact IBC@westernu.edu 909-469-5606.
From your IRBnet page: select Forms/Templates tab, then Select the Library “Western University of Health Sciences IBC Documents for Researchers” from the drop down. 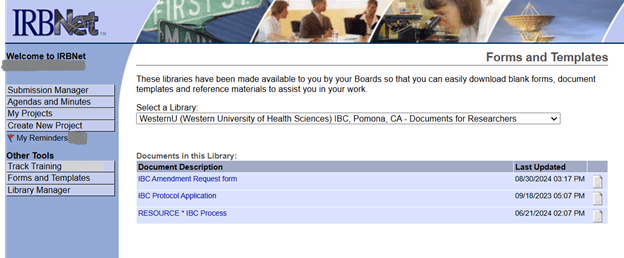
Forms Available:
IBC Application
IBC Amendment Request Form
Click on the link next to each step for a PDF with detailed instructions.
Any questions regarding registration or submission of a project to IRB, IACUC or IBC please contact Office of Research Regulatory Affairs
Step 1) IRBNet Access: www.irbnet.org
Step 2) Self Register:IRBNet Training Energizer – New User Registration (36)
- Create and Activate your account in IRBNet
- Manage affiliations from User Profile
- Add and submit necessary Training & Credential Records
- Maintain T&C records
Step 3) Create and Submit Project:IRBNet Training Energizer – Researcher 1 (32)
**Be sure to select the correct board for review. Western University of Health Sciences IACUC
- Access Forms from Forms and Templates Library
- Create New Electronic Project
- Design and Assemble Project Submission
- Share submission with Research Team
- Send project email to Research team
- Sign project package
- Submit project package for review
- Manage Projects from you My Projects Page
- Revise Incomplete Submission
Step 4) Post- Submission; Revisions, Annual Renewals:IRBNet Training Energizer – Researcher 2 (21)
- Review Project Messages and Alerts
- Create a New Package for your Project ( Revisions, Annual Renewals, Amendments)
- Add and Revise Documents
- Complete and Submit Subsequent Package
- Perform My Project Management
- Manage My Reminders
National Institutes of Health (NIH)
- NIH Requirements for IBC’s
- The NIH Process for Human Gene Transfer Trials- Frequently Asked Questions
Centers for Disease Control (CDC)
Biosafety in Microbiological and Biomedical Laboratories
Federal Select Agent Regulations:
Possession, Use and Transfer of Select Agents and Toxins
Health and Human Services Regulations:
- Interstate Shipment of Etiologic Agents (42 CFR 72)
- Etiologic Agent Import Permit Program (42 CFR 71)
Department of Labor Occupational Safety & Health Administration (OSHA)
- Occupational Exposure to Bloodborne Pathogens (29 CFR 1910.1030)
- Bloodborne Pathogens and Needle Stick Prevention
U.S. Department of Agriculture (USDA-APHIS)
- American Type Culture Collection (ATCC)
- International Air Transport Association (IATA) Guidance on Infectious Substances
- American Biological Safety Association (ABSA) Risk Group Classification
- American Biological Safety Association (ABSA) International
- California Department of Industrial Relations
WesternU IBC uses the IRBNet platform for all committee business, including agenda and minute management for all IBC meetings. For questions regarding archived IBC meeting minutes, please contact ORRA
June 12, 2025
July 23, 2025
August 27,2025
September 24,2025
October 27,2025
December 17,2025
February 25,2026

